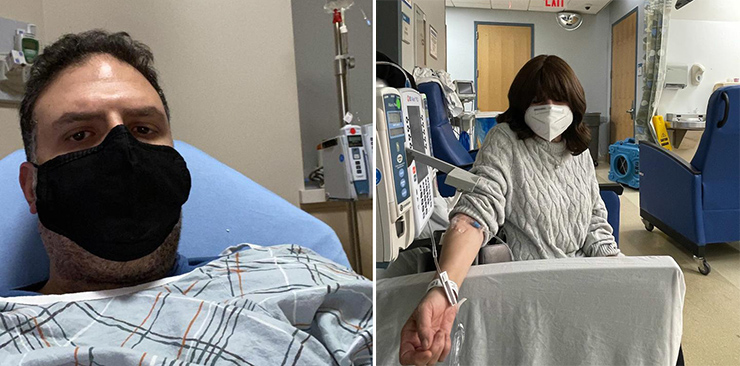
Covid Plasma Initiative’s lifesaving efforts have expanded to include the facilitation of a newly available, potentially life-saving antibody treatment referred to as monoclonal antibodies. Now in its sixth week of availability, CPI has facilitated the treatment for hundreds of people across New York, New Jersey, Chicago, Cleveland, Boston, Florida, and more.
The treatment is an infusion of lab manufactured, concentrated antibodies, similar to the antibodies present in convalescent plasma. It is the treatment given to President Trump and Governor Christie, and has been available to the public for the past six weeks.
The data thus far - and feedback from patients - is extremely positive. Covid Plasma Initiative has tracked approximately 600 NYC area high-risk patients who received monoclonal antibodies treatment in the last five weeks. Only 4 (.66%) were subsequently hospitalized for worsening symptoms. That’s in comparison to the usual hospitalization rate of >10% in high-risk patients.
CPI’s plasma collection drives have made national and international headlines and have helped countless patients hospitalized with severe COVID-19 symptoms. Now they are educating the public on the availability monoclonal antibodies; a quick and easy outpatient treatment which thus far seems to be helping people avoid hospitalizations altogether. Ads were placed this week in numerous publications across Jewish communities in the US, sharing specifics about the treatment: what it is, who qualifies, and where to go in your community to receive it.
Dr. Yosef Levenbrown is a Pediatric Critical Care Medicine Specialist at Dupont Hospital and the Medical Director of CPI (Advisory Board). He explains that the data from this double-blind randomized control trial showed that monoclonal antibodies can, potentially, reduce hospitalization significantly and decrease symptom severity.
Dr. Jeffrey Bander of Mt. Sinai Hospital states, “We’ve seen great efficacy with these drugs. As the sole outpatient treatment, monoclonal antibodies have the potential to prevent an otherwise at-risk patient from being hospitalized and keep beds available for the critically ill.”
“It’s a miracle drug,” shares Mr. Yehudah Serle (brother of Mordy Serle, one of CPI’s founders), who just recently tested positive for COVID-19 and received monoclonal antibodies. “One day I wasn’t feeling too good. I got the treatment and the next day, b’chasdei Hashem, I was feeling much better.”
Another patient, who asked to remain anonymous, thanked CPI for their assistance stating, “Just wanted to thank you for the referral to Richmond University Medical Center for monoclonal antibodies. My husband was in and out in four hours total. It was a simple and easy process and he already feels better today. He is dressed and at his desk learning Daf Yomi. Tizku l’mitzvos!”
Monoclonal antibody treatment can be accessed by anyone who tests positive and is considered “at risk.” A person is defined as “at risk” if they have one of the following factors: diabetes, obesity, immunosuppressed, over 65, or over 55 with hypertension. Other factors will qualify as well.
The FDA advises that monoclonal antibody treatment start as soon as possible after a positive test, preferably within 24 hours and no later than 10 days after symptom onset. It is therefore imperative to take a COVID test right away if you have symptoms or were exposed. Contact your doctor immediately to discuss whether this treatment is appropriate for you and if so, to arrange treatment.
Locations currently providing monoclonal antibodies include: Maimonides (Brooklyn), NY Community Hospital (Brooklyn-positive PCR test result required), Good Samaritan (Suffern), Richmond University Medical Center (Staten Island) and ODA Wallabout Health Center (by appointment only- 718-260-4600). For a complete list of all facilities providing monoclonal antibody treatment, please visit CPI’s website, PlasmaHeroes.com. (Please note that not all hospitals on this list are similarly recommended for COVID-19 inpatient care).
For more information, visit www.covidplasmasavealife.com. Contact: This email address is being protected from spambots. You need JavaScript enabled to view it./828-4PLASMA